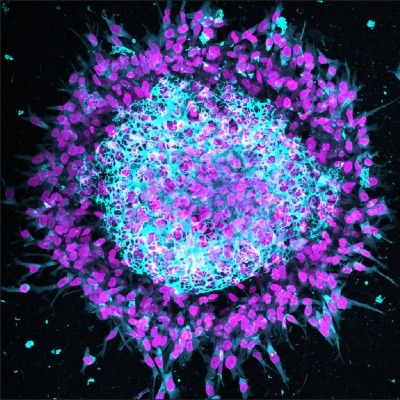
Gum cells, rather than patrolling immune cells, may be the harbingers of chronic inflammation that lead to gum disease.
NIDCR News 2024
More than 300 trainees attended NIDCR’s “Celebrating Trainees: Past, Present, and Future” Symposium as part of the institute’s 75th Anniversary celebrations. Nobel Laureate Dr. Ardem Patapoutian delivered the keynote address, and both days featured plenary sessions with NIDCR leaders. Trainees additionally had the opportunity to share and discuss their work in poster sessions, learn more about NIDCR’s grants and funding process, and receive career advice from experts during breakout sessions.
Over the last 75 years, NIDCR's investments have yielded a more complete understanding of the causes of periodontal disease and its effects on oral and overall health.
The U.S. Senate, with bipartisan support, has introduced resolutions recognizing the 75th anniversary of NIDCR.
Silver diamine fluoride can be safely, easily, and painlessly swabbed onto cavities as a potential tool for improving children’s oral health.
For 75 years, NIDCR-supported science has brought healthy smiles to children. From therapies to halt tooth decay to AI-powered cavity-detection apps, ongoing research aims to break down barriers to oral health care.
A time-lapse video shows cancer cells can sprout long appendages to force their way into healthy tissue.
Office plays vital role in institute's planning efforts.
Harold Slavkin will be remembered for his passion for craniofacial biology, his deep commitment to training future generations, and his fierce advocacy for health equity.