Itching for Answers
NIDCR scientists search for the molecules that make us itch
Why do we itch? Most of us are familiar with the nagging, squirm-inducing sensation, whether from a scratchy sweater, a bug bite, or sunburn. Usually, itch is a fleeting annoyance. But in some conditions, such as psoriasis or during healing of severe wounds, itch can become unrelenting and diminish quality of life. Existing treatments for chronic itch are not always effective. Recent research, by NIDCR scientists working in different labs with differing expertise, may help explain why. Their findings indicate that not all itch is created equal. Distinct combinations of molecules appear to underlie different types of itch, and treatments tailored to specific kinds of itch may offer more effective relief than a one-size-fits-all approach.
When the skin’s protective barrier is disturbed—by injury, an insect, or even lack of moisture from dry winter air—the immune system springs into action, mounting a defense against potential invaders and helping to heal the area. Part of this process involves the release of small proteins called cytokines, which help immune cells communicate with each other. Some of these cytokines activate itch-sensing nerve fibers in the skin. The resulting signal is relayed to the brain, where it is perceived as the sensation of itch. But scientists haven’t fully understood the exact molecules and mechanisms involved.
Scientists in the lab of NIDCR senior investigator and immunologist Wanjun Chen, MD, recently set out to determine how itch arises during wound healing. For most of us, itch is a temporary symptom that emerges as a scratch or scrape heals. But for large or severe wounds, including burns, itching can be agonizing and persistent.
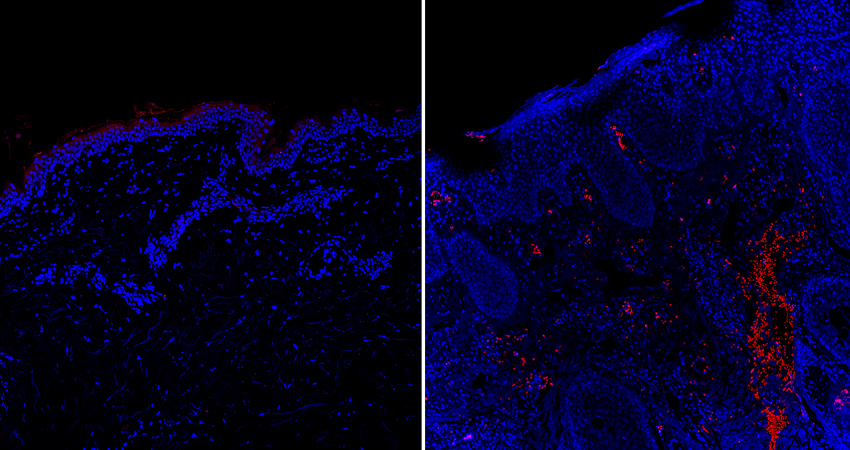
Chen and then-postdoc Junji Xu, DDS, PhD, found high amounts of a cytokine called IL-31 in skin wound tissue from mice at the peak of itch responses. In addition, wound-induced itching was eliminated in genetically engineered mice that lacked IL-31. Further experiments showed that IL-31 is released by immune cells called dendritic cells, which rush to sites of skin damage to defend against pathogens. The results pointed to an important role for IL-31 in wound-induced itching.
Although they had pinpointed the likely immune mechanisms, Chen’s team needed to understand the other side of the equation—how that activity translates into the itchy feeling perceived by the brain.
“We knew the immunological part very well, but we’re not experts in the neural mechanisms,” says Chen. So his group turned to their NIDCR colleague Mark Hoon, PhD, a senior investigator and neuroscientist who’s an expert in neurons that detect itch and pain. With help from Hoon’s lab, the scientists found that IL-31 elicits itching by stimulating itch-sensing nerves that relay signals from the wound site toward the brain.
“There’s value in venturing beyond our particular areas of expertise, because the molecular activities that occur in the skin don’t recognize the borderline between immunology and neuroscience,” says Hoon. “There’s so much at the intersection of the fields that’s really important to understand.”
Further research will be needed to determine if the IL-31 findings apply to humans. “Ultimately, the goal is to develop a therapy,” says Chen. “But IL-31 may not be the only cytokine driving itch during wound healing—there may be others too.”
Recent research from Hoon’s group appears to align with this idea. “There’s only a certain set of cytokines that are likely to be important for itch, and different combinations of cytokines may drive different itch-related conditions,” he says.
Hoon’s team looked at cytokines present in skin samples from people with chronic itch-related conditions such as psoriasis. For most, a cytokine called oncostatin M (OSM) predominated.
The researchers found that OSM enhanced the activity of itch-detecting neurons in a lab dish, and that injecting it into the skin of mice intensified scratching behaviors. In a mouse model of psoriasis-induced chronic itch, scratching was almost entirely eliminated by giving a drug that blocks OSM from interacting with itch neurons, pointing to OSM’s importance in driving chronic itch.
“Small-molecule drugs that block OSM’s activity could be promising therapies for chronic itch,” Hoon says.
But not every patient may benefit from an OSM-blocking therapy. “On average across all the patients we studied, there was very high production of OSM—but not in every individual,” says Hoon. Instead, some skin samples had high levels of IL-31, the cytokine Chen’s team found to be linked to wound itching. Further experiments by Hoon’s group showed that IL-31’s effects on itch neurons were similar to OSM’s.
“A number of cytokines could produce these effects,” Hoon says. “Ideally, if you want to treat a person most effectively, you would try to determine which molecules are elevated in their skin and target them specifically.”
With that goal in mind, the Hoon and Chen labs plan to continue identifying itch-related compounds and defining their roles, with the aim of painting a more complete picture of itch in its many forms.
“That’s the idea of personalized medicine—treating based on what’s actually happening in each patient,” says Hoon.
Related Links
References
Oncostatin M can sensitize sensory neurons in inflammatory pruritus. Tseng PY, Hoon MA. Science Translational Medicine 2021.13,619:eabe3037. doi:10.1126/scitranslmed.abe3037
The Cytokine TGF-β Induces Interleukin-31 Expression from Dermal Dendritic Cells to Activate Sensory Neurons and Stimulate Wound Itching. Xu J et al. Immunity 2020. 53,2: 371-383.e5.doi:10.1016/j.immuni.2020.06.023
Attention Editors
Reprint this article in your own publication or post to your website. NIDCR News articles are not copyrighted. Please acknowledge NIH's National Institute of Dental and Craniofacial Research as the source.
November 2024