Findings may provide insights for the field of regenerative medicine
In Brief:
- Researchers have discovered that stem cells found in continuously growing mouse incisors behave in unexpected ways and are more abundant, active, and dynamic than previously thought.
- The findings challenge previous dogma about dental renewal in rodents and could inform research on tooth regeneration in people.
Humans can’t regrow their adult teeth. But every 35 to 45 days mice wear down and completely renew their four front teeth, or incisors. This trait, which they share with all other rodents, makes mice especially useful for studying how adult stem cells regenerate tissue.
Ophir Klein, a professor of orofacial sciences, human genetics, and pediatrics at the University of California, San Francisco, is interested in organ formation in the developing embryo and how organs renew and regenerate in adulthood. Klein is one of two investigators who were the first recipients of NIDCR’s Sustaining Outstanding Achievement in Research (SOAR) award in 2015. SOAR awards provide up to eight years of stable funding to a subset of mid-career scientists called early-established investigators for ambitious, long-term research programs that have extraordinary potential. With the SOAR support, Klein has been investigating how adult stem cells drive tooth renewal in rodents, knowledge that may inform research on regenerating human teeth.
Now, says Klein, “we’ve uncovered cell behaviors that expand and challenge previous views about how the tooth system functions.” Among their findings: stem cells in mouse incisors are more abundant, active, and dynamic than previously thought. The research was published in September in Nature Cell Biology.
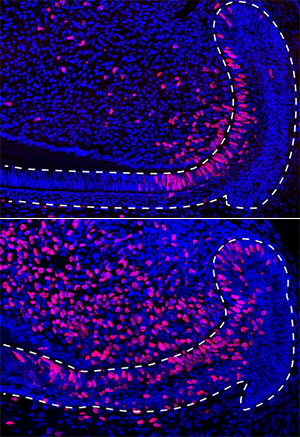
Klein and his colleagues from several institutions carefully examined the individual cells in the epithelium, a layer of the mouse incisor where stem cells give rise to one of the tooth’s main tissues, enamel. Using a technique called single-cell RNA sequencing, the team identified each cell’s unique genetic signature. Comparing gene profiles and analyzing cell changes then helped them determine how different cells in the epithelium behave. The researchers also mapped the cells to specific areas in the epithelium and showed how the dental epithelium recovers from injury.
Previously, scientists thought that the stem cells in the epithelium were not especially active and resided in its outer area. They also believed that this outer region produced both ameloblasts, the cells that deposit tooth enamel, and cells thought to provide physical and nutrient support to ameloblasts.
What Klein’s lab has shown is that the stem cells in the epithelium are actively dividing even under steady state conditions, and that they are located in the inner area of the epithelium, not the outer area. Contrary to expectations, the researchers also demonstrated that these stem cells give rise to the ameloblasts and the cells of a section of non-ameloblast epithelium that supports the ameloblasts.
To determine how the epithelium handles injury, the team damaged the tissue with a cancer drug that targets actively dividing cells. Instead of producing a cell type specific for injury repair, the injured epithelium responds unexpectedly: cells in the inner region of the epithelium become more abundant and active, and one population of support cells directly converted into ameloblasts that could then regenerate the damaged tissue.
“Although there is not a direct link between ever-growing rodent teeth and human teeth,” says Klein, “we believe that by understanding the fundamental mechanisms by which nature normally renews teeth across different types of animals, we will be able to lay a foundation for human tooth regeneration.”
Related Links
Reference
Sharir A, Marangoni P, Zilionis R, Wan M, Wald T, Hu JK, Kawaguchi K, Castillo-Azofeifa D, Epstein L, Harrington K, Pagella P, Mitsiadis T, Siebel CW, Klein AM, Klein OD. A large pool of actively cycling progenitors orchestrates self-renewal and injury repair of an ectodermal appendage. Nat Cell Biol. 2019 Sep;21(9):1102-1112. doi: 10.1038/s41556-019-0378-2. Epub 2019 Sep 2.
Attention Editors:
Reprint this article in your own publication or post it your website. NIDCR News articles are not copyrighted. Please acknowledge NIH’s National Institute of Dental and Craniofacial Research as the source.