On May 5, NIDCR issued two Notices of Special Interest highlighting the urgent need for research on coronavirus disease 2019. This research may be conducted either via the National Dental PBRN infrastructure or independently of it. The institute is prioritizing research topics that would have an immediate and high impact on the protection and safety of dental personnel and patients, including prevention of SARS-COV-2 transmission.

Dental health care personnel can find COVID-19-related information on the CDC’s Division of Oral Health website, which offers guidance and resources for clinics and health care facilities and recommendations for responding to COVID-19 in dental settings. On May 19, CDC updated its guidance for dental settings, including recommendations for resuming non-emergency dental care; expanded guidance for provision of dental care to patients with and without COVID-19; and new information regarding facility and equipment considerations, sterilization and disinfection, and test-based strategies to inform patient care.

Biochemist Nadine Samara, PhD, recently earned tenure-track status at NIDCR through the prestigious NIH Stadtman Investigators and NIH Distinguished Scholars programs. Samara is studying the structure and function of a family of enzymes called glycosyltransferases, which are key to microbial balance in the mouth and could be promising targets for treating oral microbial infections.
Researchers identified a group of neurons in mice that blunt pain by dampening the activity of multiple pain-processing regions of the brain. The neurons could be promising targets for new non-addictive therapies for chronic pain.
Scientists used a new technique to visualize fluorescently labeled bacteria in intact biofilms scraped from the tongues of 21 healthy human volunteers. They found that three types of note—Actinomyces, Rothia, and Streptococcus—were present in all participants. The prominence of these microbes suggests they may be important in maintaining oral and overall health.
Using small-molecule inhibitors that act on cell signaling, NIDCR researchers restored salivary function in mouse models of Sjögren’s syndrome. The scientists’ target was a protein called BMP6, a possible driver of salivary gland dysfunction in this autoimmune disease.
NIH’s I Am Intramural Blog reports on a study by NIDCR’s Eva Mezey, MD, PhD, who found that cells from bone marrow could be used to calm out-of-control immune cells that harm the lungs of patients with an inflammatory disease called sarcoidosis. Future studies will be needed to determine how a cell-based treatment for sarcoidosis might be delivered into patients’ lungs.

Researchers have developed fillings that are twice as strong and longer lasting than those now used in dental clinics. The next step is to make those fillings more resistant to cavity-causing bacteria without harming the beneficial species that live in the mouth.
An NIH COVID-19 microsite offers a curated collection of information and resources about the status of the outbreak, including the latest CDC information and guidance, NIH research on the novel coronavirus, information for NIH applicants and recipients, NIH guidance on travel and meetings, and a link to a portal where researchers can submit diagnostic, therapeutic, vaccine, and other candidates or technologies for cataloging by NIH. The microsite's information is updated regularly. The site also provides a link for users to sign up for COVID-19 email updates from NIH.

NIH and the Foundation for the NIH are bringing together biopharmaceutical companies, HHS partners, and the European Medicines Agency to develop a coordinated research response to the COVID-19 pandemic. The planned Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) partnership will provide infrastructure, subject matter expertise, and/or funding to help move promising vaccine and drug candidates into clinical trials.

On April 29, NIH announced the Rapid Acceleration of Diagnostics Initiative (RADx), which aims to speed innovation, development, and commercialization of COVID-19 testing technologies. NIH, working closely with the US FDA, CDC, and the Biomedical Advanced Research and Development Authority, will put rapid testing technologies through a highly competitive, three-phase selection process to identify the best candidates for at-home or point-of-care tests for COVID-19. The goal is to make millions of accurate, easy-to-use tests per week available to all Americans by the end of summer 2020, and even more by flu season. NIH urges scientists and inventors working on rapid testing technology to compete in the national challenge for a share of up to $500 million over all phases of development.

The NIAID Strategic Plan for COVID-19 Research, released April 23, outlines the institute’s strategy to accelerate research on the diagnosis, prevention, and treatment of COVID-19. The plan highlights four priorities: improvement of fundamental knowledge of SARS-CoV-2 and COVID-19; development of rapid, accurate diagnostics and assays to identify and isolate COVID-19 cases and track the spread of the virus; characterization and testing of potential treatments for COVID-19; and development of safe and effective vaccines to protect individuals from infection and prevent future SARS-CoV-2 outbreaks.

The National Library of Medicine is collaborating with publishers and scholarly societies to increase the number of coronavirus-related journal articles in PubMed Central, along with available supporting data. The articles will be in machine-readable formats that will allow artificial intelligence researchers to develop text-mining approaches to address questions about coronavirus.

CDC on May 1 announced the launch of the SARS-CoV-2 Sequencing for Public Health Emergency Response, Epidemiology, and Surveillance (SPHERES) consortium, which will greatly expand the use of whole genome sequencing of the COVID-19 virus. SPHERES will provide consistent, real-time sequence data to the public health response teams investigating cases and clusters of COVID-19 across the country. It will help them better understand how the virus is spreading, both nationally and in their local communities.
The Rare Diseases Clinical Research Network, led by NIH’s National Center for Advancing Translational Sciences, in collaboration with nine other NIH institutes and centers including NIDCR, launched a new online survey that aims to determine how the COVID-19 pandemic affects individuals with rare diseases, their families, and their caregivers. Results will help the rare disease research community shed light on the needs of people with rare diseases during the COVID-19 pandemic and other potential health crises, in addition to informing future research efforts.
A panel of experts—among them frontline clinicians—has developed COVID-19 treatment guidelines for healthcare providers. The guidelines, which will be updated as new data are published in peer-reviewed scientific literature, address treatment with antivirals, host modifiers, and immune-based therapies; patient evaluation and stratification based on infection risk and illness severity; critical care; and other topics.
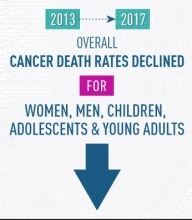
The Annual Report to the Nation on the Status of Cancer, released March 12, found death rates for most cancers have continued to decline. From 2013 to 2017, death rates for men decreased for 11 of the 19 most common cancers; among women, death rates decreased for 14 of the 20 most common cancers. During the same period, death rates for cancers of the oral cavity and pharynx increased for men and remained stable for women. The report was issued jointly by the National Cancer Institute, CDC, the North American Association of Central Cancer Registries, and the American Cancer Society.